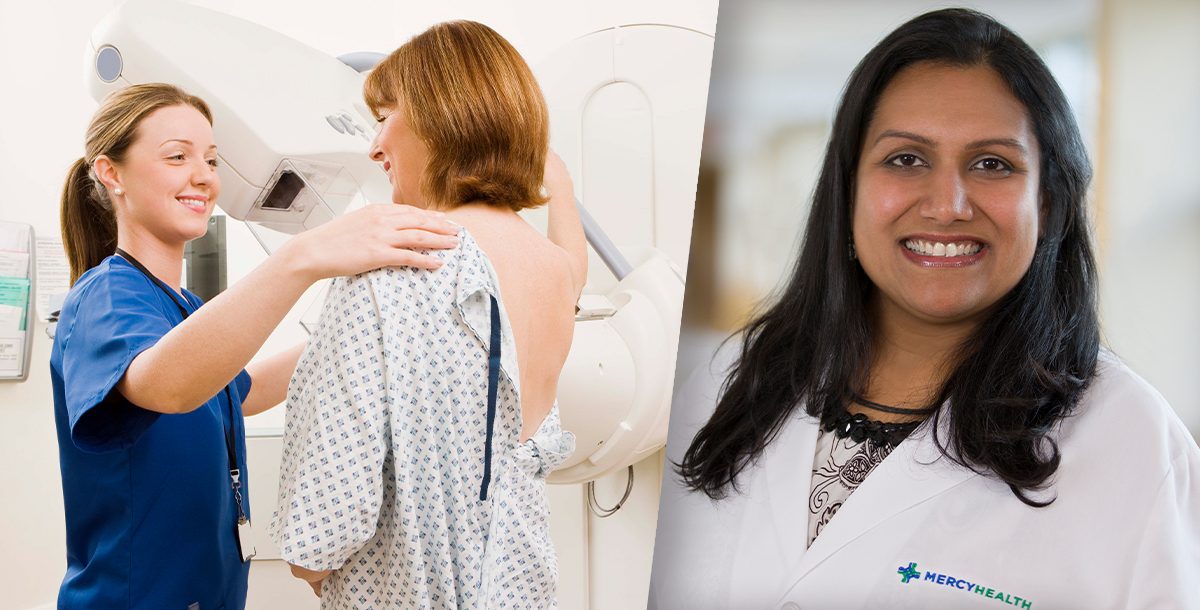
The U.S. Food and Drug Administration (FDA) recently released new rules regarding mammograms that may help more women detect breast cancer earlier. These new standards require providers to notify women if they have dense breast tissue and recommend they consult with a doctor about whether they should get additional screening.
We spoke with Mita Patel, MD, FACS, a breast surgical oncologist and medical director of our breast cancer program in our Lorain market. She explains more about what you can expect as a result of the FDA’s changes.
Can you describe the new guidelines?
“The FDA has updated the mammography guidelines by requiring mammography facilities to notify patients about the density of their breasts. Dense breast tissue may make cancer detection more difficult on mammograms. Dense breast tissue is also a risk factor for developing breast cancer. There will be further recommendations for the patient to talk to their provider about their breast health and risk factors for developing breast cancers.”
How are they different from the previous ones?
“(The updates are) making it a standard so referring providers are clear on how to communicate with their patients.”
What impact do you think these changes will make?
“Patients and providers will be aware of their breast health and can have further conversations on the risks for breast cancer.”
As a provider, will these changes help you in diagnosing breast cancer?
“Not necessarily, but (we can) have more conversations about breast cancer risk, and potentially patients may qualify for other testing modalities that might detect cancer better, such as a breast MRI.”
Are there any challenges that the new guidelines could create?
“It might create more anxiety amongst patients and a false assumption that if they have dense breasts, mammograms are not needed. (Mammograms) are still important in screening and early detection, but patients should have the conversation of their true risk and additional modalities to detect cancer.”
Can you identify any other changes that could be made when it comes to guidelines for mammograms?
“(I am) hoping they could use risk calculations in all patients so they can follow up in a high-risk breast clinic.
The amendments also help bring the Mammography Quality Standards Act (MQSA), which regulates the quality of care in mammography, into the 21st Century, including modernizing the regulations by incorporating current science and mammography best practices to improve breast cancer detection, as well as helping empower patients with more information when they are considering important decisions regarding their breast health care.”
Learn more about breast cancer as well as the mammogram and breast imaging services we provide at Mercy Health.